I was somewhat misquoted as to what I related to my friend about
the forging question. Indeed, forging will produce smaller crystals
more equitably distributed in the mass of metal. A good thing.
I copied and pasted your post so I do not see how I misquoted you
and I do not see how it was out of context either. Ant neither one
was my intention.
What I maintained is that when an ingot is cast in preparation for
milling, then annealed and put through several passes in the mill,
annealed again, rolled, etc. the results would bed similar.
Any piece of metals microstructure is a determined by its chemistry
and its work history. The results between a forged, annealed and
then rolled billet and an ingot that has just been rolled to the
same amount of reduction will differ. With enough rolling and
annealing cycles they will become virtually indistinguishable but in
precious metals we don’t often make ingots large enough to
accomplish that much work. Certainly at the level of a studio smith
you might do two cycles of proper rolling and annealing to get to
final working thickness but not much more. So in that case it would
be still apparent what the difference was.
This is what we do and have not seen the failures mentioned by
those who advocate forging before rolling. My understanding is that
the function of annealing is to change the crystalline structure so
that stress leads to "slippage" between crystals without
stress-linked fractures.
What annealing does if there was an adequate amount (typically
greater than 50 % reduction in section) of cold work prior to
annealing is to return the crystal matrix to a relaxed state and if
there was enough cold work then there will be recrystallization
where the energy required to form new crystals is lower than the
energy for the crystal to morph back its equiaxed shape. This
recrystallization is what makes the smaller equiaxed crystal
lattice.
Slippage is along planer layers of atoms in the lattice of a crystal
creating slip planes. Grain or crystal boundaries are impediments to
slip as the orientation of planes in adjacent crystals is different
so the direction of slip will need to be different requiring more
energy. This is what makes poly crystalline materials stronger than
single crystals and why many small equiaxed crystals will be a
durable configuration than larger or more directional oriented
crystal lattices. If there is enough strain on a pair of adjoining
crystals they will not deform by internal slip but fracture, larger
or longer crystal boundaries allow the matrix to fracture more
easily as the longer boundaries more easily propagate the fracture.
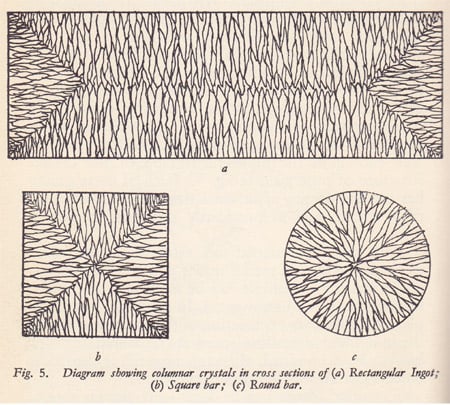
Which brings us back to the ingot. look at this image of the crystal
structure of an as cast ingot
http://www.ganoksin.com/gnkurl/ep7zec
In a metal ingot mold as the molten metal is introduced the mold it
begins to immediately nucleate crystals on the mold wall due to the
metal molds high thermal conductivity and high thermal diffusivity.
This produces a layer of very fine tight equiaxed “chill” crystals
then as the thermal flux is reduced by this layer the faces of those
chill crystals pointing in a favorable direction into the melt begin
to grow into long columnar crystals oriented towards the center of
the molten volume. Ideally there will then be enough reduction in
heat transfer for the middle of the ingot to slow its cooling enough
for more equiaxed grains to begin to precipitate out of the melt
resulting in the structure shown in that URL.
Depending on the amount of super heat (heat in excess of the melting
temperature) in the metal and melt/ingot size those columnar grains
can continue all the way to the middle of the ingot this will create
what is known as a pipe and can lead to significant problems when
rolling as the impurities in the melt will mostly end up where the
advancing columnar grains meet from the opposing and adjacent sides
leading to a weakness in the crystal lattice. This is the common
structure fro the ingots that we cast as studio smiths because we
have tiny melt volumes and we are struggling to get enough heat into
the melt to keep it from freezing It looks more like this.
and this image of a silver ingot interior

The ingots will have a significant tendency to fail at the
intersections of the columnar grains from the faces of the mold. So
combining this with the long perpendicular grain boundaries of the
columnar grains and strain introduced by the shrinkage of
solidification you have a piece of metal that is not in optimal
condition for rolling. Will this structure automatically result in
failure, no it will not but it will increase the likelihood of
defects in rolling and further on down the line.
Bu subjecting the as cast ingot to heavy forging those structures
will be destroyed by the deformation and subsequent annealing to
produce a billet with an fine equiaxed grain structure that will
better stand up to further deformation in rolling and other forms of
plastic deformation in later processing stages.
I admit that my academic and practical experience is more
applicable to steel and its alloys. I think the principles are
largely the same. In steel (0.25% carbon) heating above 1350 F.
ferrite recrystallizes from alpha to gamma form (austenite). Upon
cooling the final grain size of pearlite and ferrite depends
greatly upon the fates of heating and cooling.
( I know that there are other crystal forms such as the hard
martensite and cementite, but lets leave it here). Cool too fast
and the steel is too hard to work easily with a hammer. Held at the
right temperature and cooled at the right speed, annealing occurs.
Steel is a much more complex animal due to its interstitial solid
solution crystal structure it forms many complex crystal states
depending on temperature and speed of heating and cooling. Most of
the alloys we are concerned with are substitution solid solutions
with much simpler behavior.
http://www.ganoksin.com/gnkurl/ep7zed
My understanding of how silver (which I prefer to use in most
cases for jewelry) and gold respond to annealing is that annealing
will mitigate fractures when the metal is worked mechanically up to
the point where it is work hardened and will break when deformed.
If the lattice is actually stressed to the point of fracture
annealing will not fix it. With a fracture the only way to fix it if
it doesn’t actually come to the surface is hot forging or Hot
Isostatic Pressing then you may be able to get diffusion to fix the
crack.
So, what I said to my friend was that forging would alter
crystalline structure. It is counterintuitive to me that a forged
piece of sillver or gold will behave better when rolled than ones
that have simply been annealed to survive the mechanical stress of
milling which would seem to be similar to forging. I did not think
that the forging was necessary unless it solved some problem
(which we are not having).
Forging and rolling apply deformation to the metal in different ways
rolling presents a much higher stress to the metal. Forging delivers
more of its energy to the area below the struck surface because
friction keeps the material in direct contact with the hammer and
anvil from spreading sideways. So in effect it works the interior of
the metal more than the exterior surfaces as shown here
http://www.ganoksin.com/borisat/nenam/nenamart/brepohl-clipart/4-44.gif
In rolling the stress is distributed more like this
http://www.ganoksin.com/borisat/nenam/nenamart/brepohl-clipart/4-46.gif
Unless you have a very stout powered mill with large diameter rolls
the middle shaded area in that diagram is not affected by rolling
and you end up with a very unequal distribution of stresses across
the whole width and length of the rolled sheet. The this article
that these images came from is the section of Chapter 4 in Brephol
that covers much of what we have been discussing here and much to my
surprise I found it here on Ganoksin today while looking for images
to illustrate this post, thanks to Brynmorgen Press and Hanuman for
publishing it there.
The second problem I found in the discussion was the fact that
there seemed to be no quantification in the statements. The forged
piece will last longer---years, decades, centuries??? How much
longer (I suspect that this cannot be answered and wont expect an
answer but would be delighted to get one.)
I am in agreement with you here in that there is no data I know of
to quantify this.
Further, at one point we are told that "a lot of small taps is
much better than fewer hard taps". I do not automatically
understand the value of "a lot", "small taps", etc. I suppose
watching you doing the job would help, but I suspect that the force
of a hammer blow is strongly affected by ones arm and the hammer
handle length and weight of the head.
As I did not make this assertion I cannot make any comment on it. I
tend to use power hammers and presses to forge my ingots.
The metal hammered pre-milling is denser. This is likely to be
true--at first. Is this true after the metal is worked by milling?
Is this one of the endless and mainly illogical discussions such
as whether a cast piece can be hand made? Honestly, I don't know
and haven't found the to help me decide.
The metal cannot be made denser in any practical sense by forging or
rolling. Density is a physical property of the metal that short of
terribly extreme forces (think nuclear explosion or black holes) we
cannot change. I cannot imagine an ingot could contain enough gas
porosity to significantly effect its density and if it did it would
likely exhibit severe defects during processing that would make the
density issue moot. SO no forging or rolling is not going to change
density.
It is obvious that I am not a metallurgist, but this stuff is
neither rocket science not politics. There should be a reasonable
answer
I hope some of what I have written here will help provide some
answers. I have not written more lately due to shattering and
subsequent surgery on my right index finger which has made typing
most cumbersome. It is finally getting to the point where I am able
to type longer posts.
Regards,
Jim
James Binnion
James Binnion Metal Arts

